Europe is rising as a frontrunner within the realm of high quality management (QC) for cell and gene remedy manufacturing, paving the best way for vital developments within the discipline. Europe’s dedication to making sure the protection, efficacy, and high quality of this revolutionary remedy is enjoying a pivotal function.
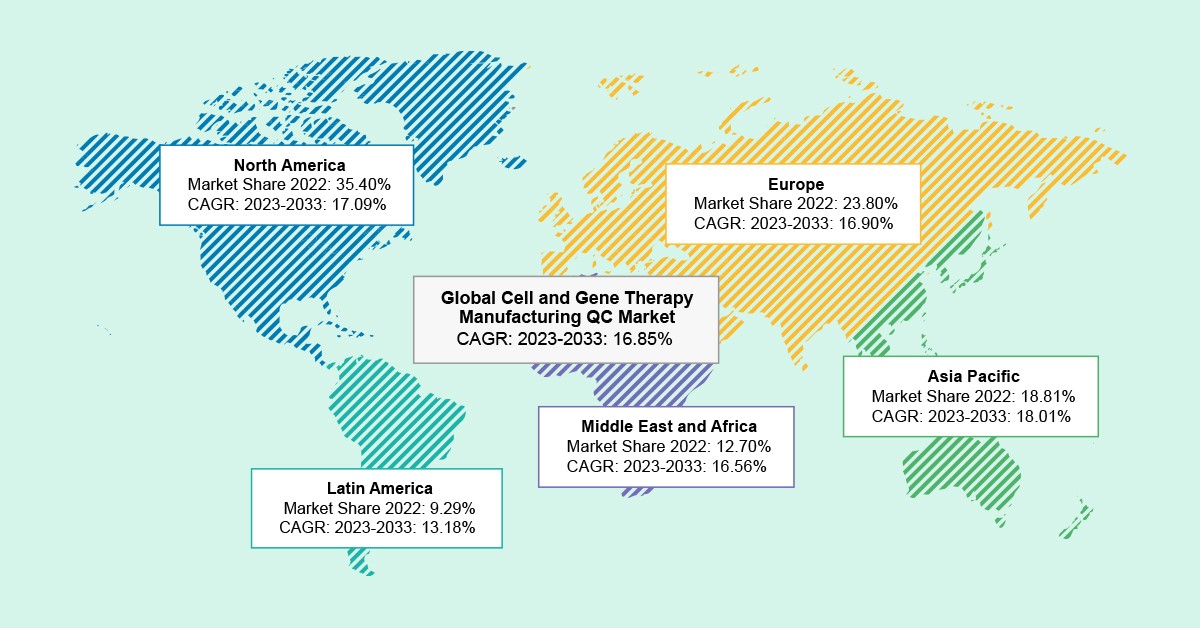
As per the BIS Analysis report, the world cell and gene remedy manufacturing QC market was valued at $1.95 billion in 2022 and is anticipated to succeed in $10.65 billion by 2033, witnessing a CAGR of 16.85% through the forecast interval 2023-2033.
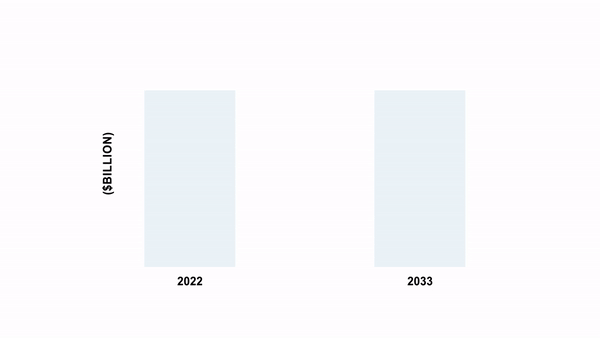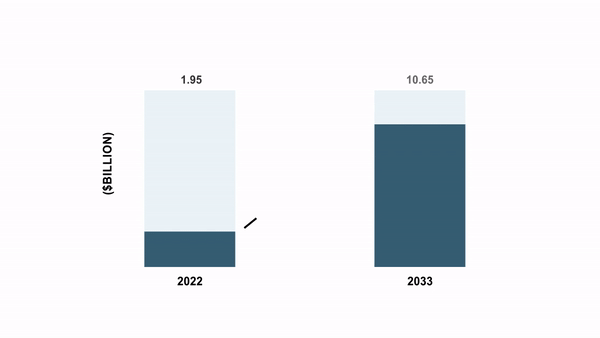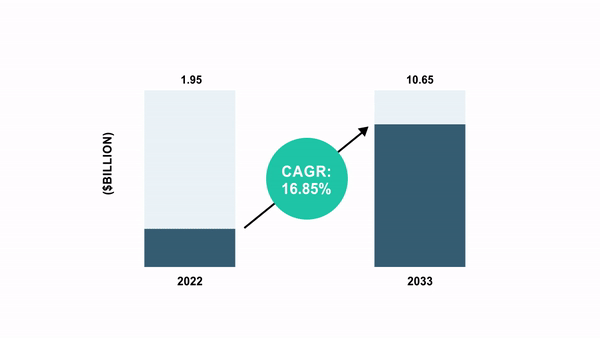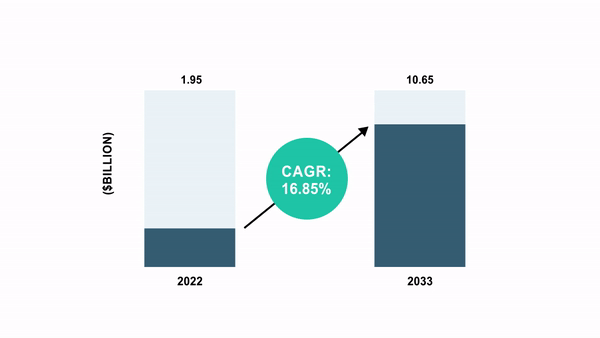
Discover extra particulars on this report on this FREE pattern.
Europe isn’t solely bolstering affected person confidence but additionally fostering a positive setting for the analysis, growth, and commercialization of this transformative remedy.
Dynamic Affect of High quality Management in Cell and Gene Remedy Manufacturing in Europe
The U.Okay. held the biggest market share within the cell and gene remedy manufacturing QC market in Europe, representing 35.07% in 2022. Moreover, it’s anticipated that the QC in France will expertise a development price of 18.67% from 2023 to 2033, indicating vital enlargement inside the forecast interval (2023- 2033).
Europe’s dedication to advancing the sphere of therapeutic analysis and growth is clear by means of its elevated funding within the sector. This dedication is essentially pushed by the presence of outstanding firms, together with Sartorius AG, Merck KGaA, bbi-biotech GmbH, and Endress+Hauser Group Companies AG (Analytik Jena GmbH).
These are famend gamers within the world business for offering high quality management options in cell and gene remedy manufacturing. By harnessing the experience of those established firms, Europe is poised to solidify its place as a frontrunner in pharmaceutical manufacturing.
Latest Developments in Europe’s CDMO Sector for High quality Management
SCTbio, a contract growth and manufacturing group (CDMO), and Cyto-care.eu GmbH, a specialised firm in superior options for cell remedy and regenerative drugs, entered right into a collaborative settlement in June 2023 geared toward enhancing the standard of cryopreservation for cell remedy development in Europe.
By this collaboration, the businesses search to deal with the demand for streamlined processes encompassing the gathering, cryopreservation, high quality management, and logistics of leukapheresis supplies, thereby supporting completely different elements of the cell remedy workflow.
Moreover, Recipharm, a outstanding CDMO, executed a collection of focused acquisitions to boost its biologics portfolio by welcoming esteemed business specialists Arranta Bio, GenIbet, and Vibalogics into its ecosystem. That is notable for the event and manufacturing of revolutionary merchandise equivalent to oncolytic viruses, gene therapies, and vaccines, contributing to the progress of human well being.
Conclusion
Thus, from sturdy regulatory frameworks to cutting-edge analytical applied sciences, Europe’s proactive strategy positions it as a key participant in shaping the way forward for the cell and gene remedy panorama.
to know extra concerning the creating applied sciences in your business vertical? Get the newest market research and insights from BIS Analysis. Join with us at hiya@bisresearch.com to be taught and perceive extra.














